2.2 Water
Understandings:
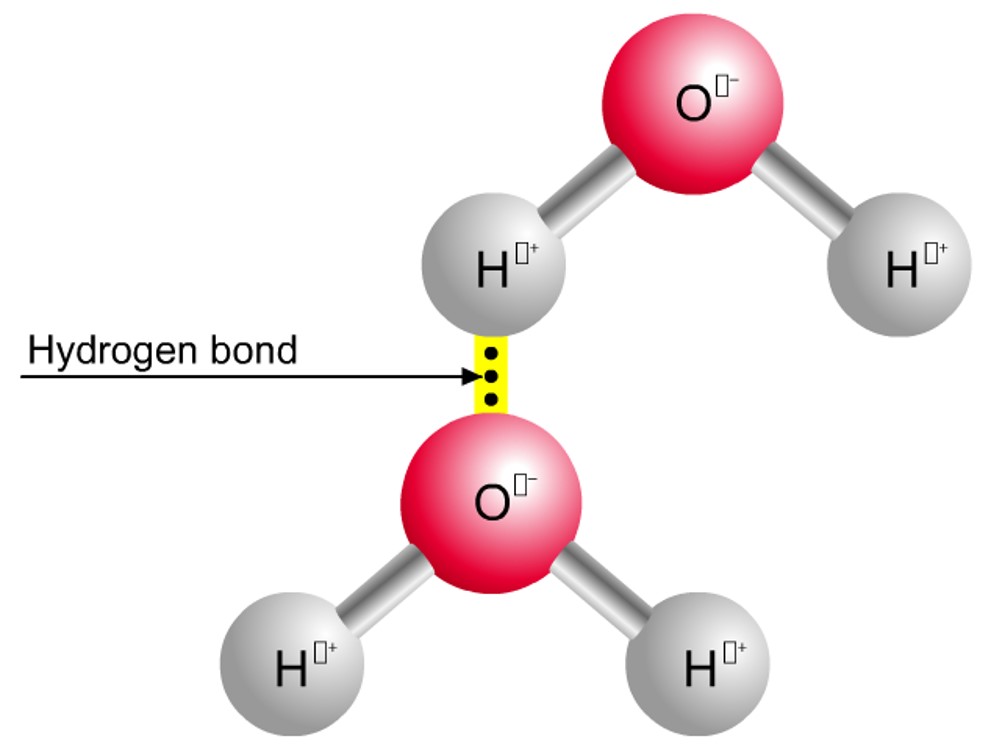
• Water molecules are polar and hydrogen bonds form between them.
• Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
• Substances can be hydrophilic or hydrophobic.
Applications and skills:
• Application: Comparison of the thermal properties of water with those of methane.
• Application: Use of water as a coolant in sweat.
• Application: Modes of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
• Water molecules are polar and hydrogen bonds form between them.
• Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
• Substances can be hydrophilic or hydrophobic.
Applications and skills:
• Application: Comparison of the thermal properties of water with those of methane.
• Application: Use of water as a coolant in sweat.
• Application: Modes of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
Full online tutorial on water: |
In Our Time Radio 4 programme on Water |
Homeopathy (ToK)
|
|
A little more serious...
|